Sample providing
Each sample and associated data request is reviewed by the BBM 1FM CU Approvals Committee individually on the basis of following criteria:
- The credibility and competence of the requester and the requester's institution
- Approval of the project by the Ethics Committee of the relevant institution
- Use of samples / data for research specified in the donor's consent
- Availability of samples
- The proposed use of samples / data consistent with the BBM 1FM CU aims
- Potential scientific impact of the project (high / low)
- The project complies with scientific standards
- Rarity of required samples
- Focus of the requester: profit / non-profit
- Type of analyses performed on the requested samples (including protocol)
- Suitability of required samples for the proposed research
- Sufficiency / redundancy of the required sample quantity
- Research project is not repeated by other requestors
Sampling
The samples are mostly provided for BBM 1FM CU in cooperation with the General University Hospital in Prague (VFN Praha). They are obtained on the basis of the signed consent of the sample donor or close person. Samples are obtained from patients with various types of diseases, especially cancer and rare diseases during scheduled examinations or surgical interventions. Samples from healthy donors (control samples) are also collected, most often during planned examinations or withdrawals. Samples from deceased donors are obtained based on the consent of a close person.
The most common types of tissue, blood and urine are obtained. The sample is collected, processed and stored according to valid SOPs of the BBM 1FM CU.
Sample processing
 The samples are obtained and processed according to the current SOPs of the BBM 1FM CU. The time between sample retrieval and aliquot preparation is given by the SOPs. All sample processing data are recorded in the biobank’s database. The presence and quality of tumour tissue is confirmed in the samples taken by the pathologist either from a frozen section or by a final examination.
The samples are obtained and processed according to the current SOPs of the BBM 1FM CU. The time between sample retrieval and aliquot preparation is given by the SOPs. All sample processing data are recorded in the biobank’s database. The presence and quality of tumour tissue is confirmed in the samples taken by the pathologist either from a frozen section or by a final examination.
The following types of samples are prepared:
Tissue:
- frozen tissue: aliquots measuring 2-3 x 4 x 7 mm
- tissue preserved in RNA later: aliquots measuring of 2-3 x 4 x 7 mm
DNA:
- isolation from whole blood by manual salting method or automatic analyser
Blood:
- Whole blood
- Serum - aliquots of 0.5 ml
- Plasma - aliquots of 0.5 ml
Urine:
- Aliquots of 0.5 ml
Liquor:
- Aliquots of 0.5 ml
Sample storage
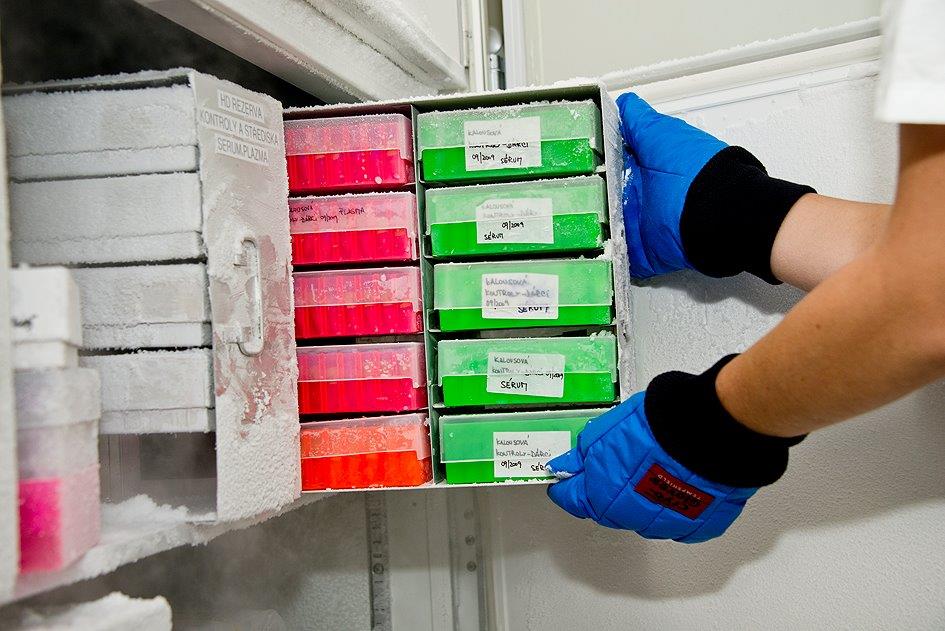
BBM 1FM CU stores samples and archives the accompanying data according to valid standard operating procedures (SOPs). The main goal is to maintain high quality samples. Individual storage data (number of aliquots, position in the appropriate storage device) are recorded in the biobank’s database.
BBM 1FM CU uses the following storage methods:
- Storage in freezers at -80 °C (blood, serum, plasma, peripheral blood DNA, urine, liquor, tissue samples in RNA later)
- Storage in vapor of liquid nitrogen at -156 °C (tissue samples)
Services for external entities
- Providing samples for science, research and teaching purposes
- Ensuring collection, preparation and storage of samples
- Storage of samples
- Isolation and processing of nucleic acids
- Preparation and staining of histopathological sections
- Finding above-standard data for samples, consultation regarding the quality of samples
- Consultation of ethical issues



